1. Evolutionary response to climate change
Global warming will change the growing season in many parts of the world. In temperate areas frosts will stop sooner in spring and start later in fall. In arid areas, growing season will contract with drought. These changes in the seasonal rhythms will alter the selective regimes acting on the genes that control when, and for how long, plants come into flower.
When the shift from vegetative growth to reproduction occurs too early, plants have few resources to make progeny. When the shift is too late they do not have enough time to mature their progeny. My lab is investigating this trade-off using field mustard, Brassica rapa, a winter annual.
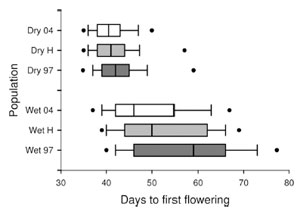
Over the early 2000’s, decreased spring precipitation in California abbreviated the growing season for B. rapa. My lab detected a rapid evolutionary shift towards earlier flowering using a protocol we call the “resurrection paradigm.” Seeds collected in 1997 (before drought) and held in cold storage were revived and grown side-by-side with seeds collected from the same populations in 2004 (after 7 years of drought). Given the same environmental conditions, the post-drought generation flowered 16% sooner than the pre-drought.
Based on this success, I initiated Project Baseline. This initiative has collected, dried and frozen seeds from multiple wild populations of 65 plant species. As global warming proceeds, these ‘ancestral’ seeds can be resurrected and grown beside their ‘descendents’ to monitor the rate of evolutionary change in a variety of functional characters.
Key Publications:
Franks, S.J., Sim, S., & Weis, A.E. (2007). Rapid evolution of flowering time by an annual plant in response to a climate fluctuation. Proceedings of the National Academy of Sciences 104:1278-1282 (pdf)
Etterson, J. R., Franks, S. J., Mazer, S. J., Shaw, R. G., Gorden, N. L. S., Schneider, H. E., Weber, J. J., Winkler, K.J., & Weis, A. E. (2016). Project Baseline: An unprecedented resource to study plant evolution across space and time. American Journal of Botany, 103:164-173 (pdf)
2. Assisted Migration and Assisted Gene Flow as Conservation Strategies.
 As climate envelopes shift poleward, narrowly adapted species may decline as the temperatures rise about them. One proposed remedy is Assisted Colonization, that is, managed establishment of new populations beyond current range boundaries. A related remedy is Assisted Gene Flow, whereby individuals are moved between existing populations to increase the local genetic variance, the prerequisite for adaptive evolutionary responses.We are evaluating the feasibility and pitfalls involved in these remedies in experiments that mimic the initial stages of an AM and AGF programs. Several projects use the Experimental Climate Warming Array at the Koffler Scientific Reserve. This field installation applies infrared warming (2C above ambient) to replicated field plots, thus mimicking mid-century climate projections.
As climate envelopes shift poleward, narrowly adapted species may decline as the temperatures rise about them. One proposed remedy is Assisted Colonization, that is, managed establishment of new populations beyond current range boundaries. A related remedy is Assisted Gene Flow, whereby individuals are moved between existing populations to increase the local genetic variance, the prerequisite for adaptive evolutionary responses.We are evaluating the feasibility and pitfalls involved in these remedies in experiments that mimic the initial stages of an AM and AGF programs. Several projects use the Experimental Climate Warming Array at the Koffler Scientific Reserve. This field installation applies infrared warming (2C above ambient) to replicated field plots, thus mimicking mid-century climate projections.
Some initial findings show that translocating populations across latitudes is prone to failure. Although southern plants have the heat tolerance genes favored when norther locations are warmed, this advantage is wiped out by their genes for flowering time. The exaggerated photoperiodic cycle at upper latitudes triggers flowering too late for southern plants; they are killed by frost before they set seed. This precludes establishment in the case of AM, and impedes introgression into the local gene pool in the case of AGF.
More recently we used Brassica rapa as a model system to develop quantitative methods for use in AGF programs. These estimate the degree of phenological mismatch between populations in decline and potential migrant source populations.
Key Publications:
Wadgymar, S., M.N. Cumming & A.E. Weis. (2015). The success of assisted colonization and assisted gene flow depends on phenology. Global Change Biology 21:3786-3799 (pdf)
Wadgymar, S.M., & A.E. Weis (2017). Divergent reproductive phenology impedes Assisted Gene Flow. Conservation Biology. DOI: 10.1111/cobi.12877
3. Selection of Phenology through the Seasonal and Social Environments.

Do the genes governing flowering time have their greatest fitness effect by governing the amount of time allocated to vegetative growth, and hence plant condition at flowering onset? Or, alternatively, do these genes maximize fitness by putting flowering plants into the slice of the temporally shifting seasonal environment that best facilitates pollen transfer and fruit maturation? There is an inherent correlation between age at flowering (determines condition) and Julian date at flowering (determines environment) that must be teased apart to estimate their independent effects.
We recently completed experiments that broke this correlation by staggered planting dates for genetic strains of Brassica rapa. This produced experimental populations in which flowering age and date were positively-, negatively-, or un-correlated. The number of seed sets (female function) increased on plants that flowered at an early date, regardless of age. The number of seeds sired (male function) increased for plants that flowered at a young age, regardless of date.
Another study looked at selection through the seasonal and social environments on germination time. In this case, germination date determines which slice of the temporal environment the plant inhabits. But it also determines what type of plants it competes against. Late-emerging seedlings get overshadowed by earlier neighbors but not vise versa. We used this situation to demonstrate novel quantitative methods to partition phenotypic selection differentials into ‘hard’ and ‘soft’ components.
Key Publications:
Austen, E.J. and A.E. Weis. (2015). What drives selection on flowering time? An experimental manipulation of the inherent correlation between genotype and environment. Evolution 69:2018-2033 (pdf)
Weis, A.E., K.M. Turner, B. Petro, E.J. Austen & S. Wadgymar. (2015). Hard and soft selection on phenology through seasonal shifts in the general and social environments: a study on plant emergence time. Evolution 69:1361-1374 (pdf)